Simple Chemical Experiments
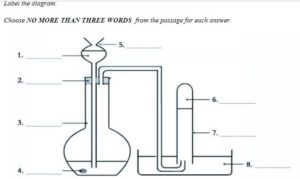
Hydrogen can be made very easily by using simple laboratory equipment. All you need is a thistle funnel, cork, glass dish, graduated glass cylinder, test tube, glass pipe, zinc, tap water, and hydrochloric acid. To begin with, 5ml of tap water is put into a 50ml graduated glass cylinder with about 1gm of zinc. The top of the glass cylinder is fitted with a cork and a thistle funnel and a glass pipe inserted into it. The glass pipe connects the cylinder with a glass dish. Sufficient tap water is placed in the dish to cover the top of the pipe which is then covered with a water filled test tube. Before adding 5 ml of hydrochloric acid to the funnel it is important to make sure that the end of the funnel is below the tap water. Once added, the hydrochloric acid comes into contact with the tap water very quickly and an immediate chemical reaction can be seen. Hydrogen gas is released and starts to travel down the pipe into the test tube. The hydrogen displaces the water and produces a test tube of pure hydrogen gas. By adding baking soda to the cylinder the acid solution is neutralized and can be poured down the sink.
Questions 1 – 8
The diagram below shows how hydrogen can be made using simple laboratory equipment.
Label the diagram.
Choose NO MORE THAN THREE WORDS from the passage for each answer.
Get your Reading eBook to learn and practice different types of questions

